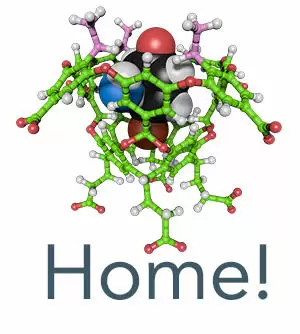
Excited-state behavior of eight α-alkyl dibenzyl ketones (alkyl = CH3 through n-C8H17) that are capable of undergoing type II and/or type I photoreactions has been explored in isotropic solution and within a water-soluble capsule. The study consisted of two parts: photochemistry that explored the excited-state chemistry and an NMR analysis that revealed the packing of each guest within the capsule. The NMR data (COSY, NOESY, and TOCSY experiments) revealed that ternary complexes between α-alkyl dibenzyl ketones and the capsule formed by two cavitands are kinetically stable, and the guests fall into three packing motifs modulated by the length of the α-alkyl chain. In essence, the host is acting as an external template to promote the formation of distinct guest conformers. The major products from all eight guests upon irradiation either in hexane or in buffer solution resulted from the well-known Norrish type I reaction. However, within the capsule the excited-state chemistry of the eight ketones was dependent on the alkyl chain length. The first group consisted of α-hexyl, α-heptyl, and α-octyl dibenzyl ketones that yielded large amounts of Norrish type II products within the host, while in solution the major products were from Norrish type I reaction. The second group consists of α-butyl and α-pentyl dibenzyl ketones that yield equimolar amounts of two rearranged starting ketones within the capsule (combined yield of ca 60\%), while in solution no such products were formed. The third group consisted of α-methyl, α-ethyl, and α-propyl dibenzyl ketones that within the capsule yielded only one (not two) rearranged starting ketone in larger amounts (21−35\%) while in solution no rearrangement product was obtained. Variation in the photochemistry of the guest within the capsule, with respect to the α-alkyl chain length of the guest, highlights the importance of how a small variation in supramolecular structure can influence the selectivity within a confined nanoscale reactor.
Publications by Year: 2008
2008
Gibb, C.; Sundaresan, A. K.; Ramamurthy, V.; Gibb, B. C. Templation of the Excited-State Chemistry of α-(n-Alkyl) Dibenzyl Ketones: How Guest Packing within a Nanoscale Supramolecular Capsule Influences Photochemistry. Journal of the American Chemical Society 2008, 130, 4069-4080.
Podkoscielny, D.; Philip, I.; Gibb, C.; Gibb, B. C.; Kaifer, A. E. Encapsulation of Ferrocene and Peripheral Electrostatic Attachment of Viologens to Dimeric Molecular Capsules Formed by an Octaacid, Deep-Cavity Cavitand. Chemistry: A European Journal 2008, 14, 4704-4710.
Giles, M. D.; Liu, S.; Emanuel, R. L.; Gibb, B. C.; Grayson, S. M. Dendronized supramolecular nanocapsules: pH independent, water-soluble, deep-cavity cavitands assemble via the hydrophobic effect.. Journal of the American Chemical Society 2008, 130, 14430-1.
At neutral pH, dendronized deep-cavity cavitands were shown to form supramolecular nanocapsules via assembly around a range of guest molecules.
Srinivasan, K.; Gibb, B. C. Synthesis of nanoscale carceplexes from deep-cavity cavitands. Chem. Commun. 2008, 4640-4642.
Deep-cavity cavitands were shown to undergo carceplex reactions in which two cavitand tetrols were covalently linked using bromochloromethane; the efficiency of the “dimerization” was closely tied to the templating guest molecule incarcerated within the host.
Ewell, J.; Gibb, B. C.; Rick, S. W. Water Inside a Hydrophobic Cavitand Molecule. The Journal of Physical Chemistry B 2008, 112, 10272-10279.
Srinivasan, K.; Laughrey, Z. R.; Gibb, B. C. Broad Functionalization of Deep-Cavity Cavitands by Directed ortho Metalation. European Journal of Organic Chemistry 2008, 2008, 3265-3271.
Abstract The endo- and exo-rim positions of deep-cavity cavitand 1 were functionalized by directed ortho metalation (DoM) procedures. A range of electrophilic quenchers led to hosts with ester, phenol and thioether functionality. In each case, a combination of host pre-organization and reaction control resulted in far fewer products than the theoretical sixty-nine. In addition to the nature of the lithiate, functionalization patterns were also dependent on the electrophile, with a general trend that the non-carbon electrophiles examined gave higher degrees of substitution. (© Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, Germany, 2008)